|
Yes, I know. And I promise you the title is not a liar. Making a gem of a tiny crystal: Slowly cooled DNA transforms disordered nanoparticles into orderly crystal
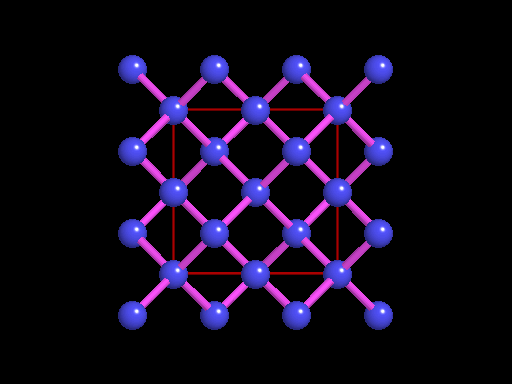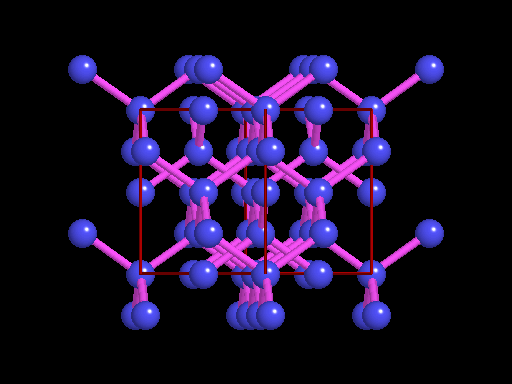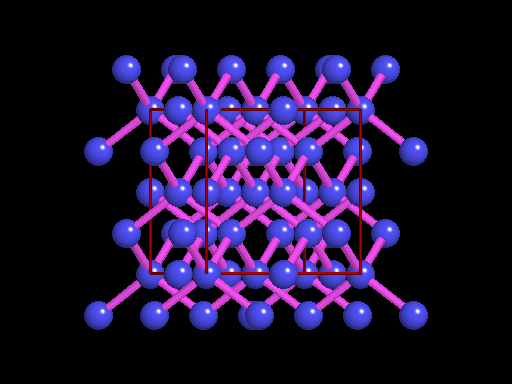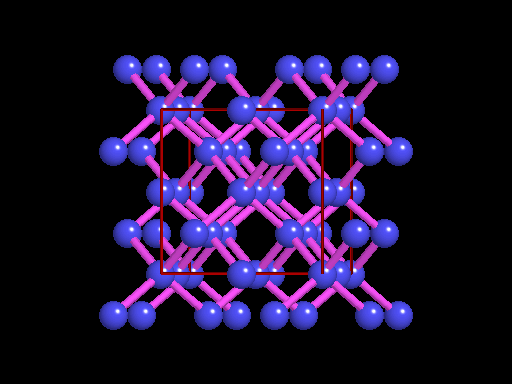
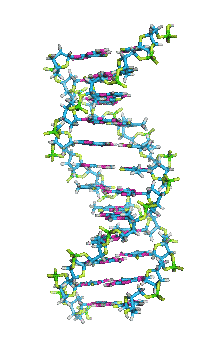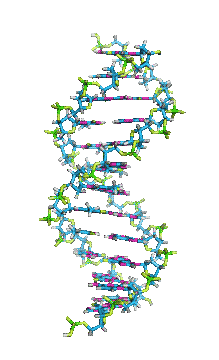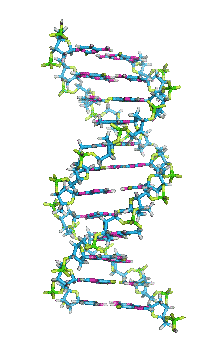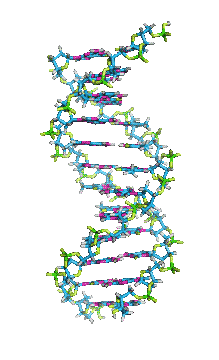
This is an example of a 3-D crystalline shape. Observe how its unique shape is derived from the way its edges and flat sides combine to form the structure. Here is a molecular model of the diamond's crystalline structure. You can see the reason why this gem possesses its specific properties; the entire shape is stacked in a very organized manner. See, nature is a beautiful thing because it has the ability to produce stunning gems like emeralds, diamonds and peridots, making humans believe that these precious minerals could only be harvested from the planet. However, times have changed. A research team in a Northwestern University has cracked the code. They've managed to successfully build the first almost-perfect crystal from nanoparticles and DNA by mimicking the same structure that nature itself has designed. Though before I move on, it is crucial you understand that DNA is literally the key to life; it contains all the secrets and mysteries of the genetic information for anything that's alive. As a result, the fact that these scientists were able to assemble an actual crystal using DNA is incredibly innovative.
 Chad A. Mirkin http://www.chemistry.north western.edu /images/ faculty/core/core-profiles/chad-mirkin-186x232.jpg Chad A. Mirkin http://www.chemistry.north western.edu /images/ faculty/core/core-profiles/chad-mirkin-186x232.jpg Nanoscientist Chad A. Mirkin explains that single crystals are the backbones, the bases of many, many things essential to our daily lives such as diamonds for jewelery and laser applications with sapphires. It's really the method in which atoms are arranged within a well-defined lattice that determines and creates these valuable crystals. I suppose you could treat these crystals as stem cells - they can be used to become anything our hearts desire. Take a moment to analyze this. What could happen if this experiment leads to a potential industry? The probability exists that it could explode with billions of dollars as it grows aggresively. Silicon, for example, is a rather important semiconductor for devices like computers and other electronics. Imagine all the benefits for novel technologies and the development of new industries if the research group is able to grow, yes grow clones of flawless crystalline structures of silicon. Brilliant, isn't it?
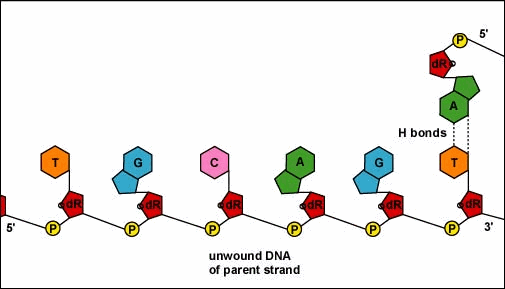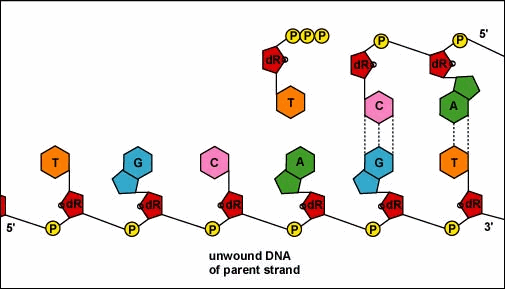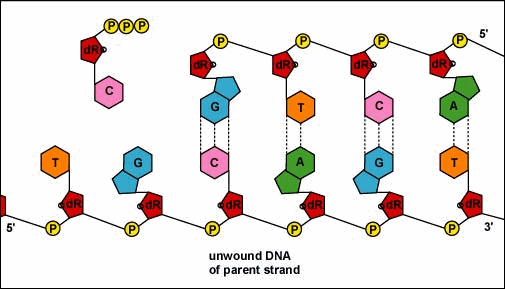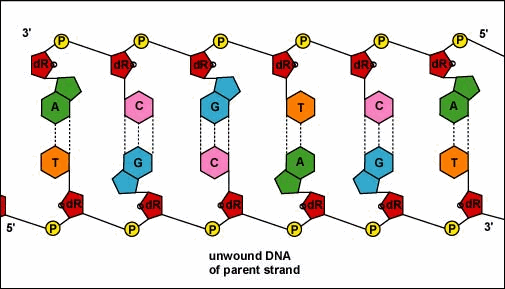
Mirkin's research team, admirably, found a way to use nanomaterials as atoms, DNA as bonds and heat to form mini crystals by fashioning a sort of "recipe". The procedure applies superlattice techniques Mirkin's team has been investigating for decades. His commitment and dedication to this project should most definitely be appreciated and applauded for; his discoveries could very much prove worthy of all the time he invested in this. To help you see my statement even more clearly, let me tell you that I haven't even existed as long as these scientists have been studying this topic. Wow. I don't know about you, but I've never felt so young before. Check out this quick video if you're interested (or even if you're confused) about crystal lattice structures  Rose quartz mineral in crystal clusters http://whataearth.com/ wp-content/uploads/2013/ 12/MB96L3.jpg Rose quartz mineral in crystal clusters http://whataearth.com/ wp-content/uploads/2013/ 12/MB96L3.jpg Most recently, Mirking has teamed up with Monica Olvera de la Cruz, a partnership between a experimentalist and theoretician respectively, to evaluate in extension the current techniques. Olvera de la Cruz suggested that in order to very accurately predict the 3-D structure (crystal shape), it all depends on the disordered set of nanoparticles and type of DNA being used. Currently, the "general" instructions provides researchers and experimentalists groundbreaking control over the types and shapes of crystals that can be manufactured. As far as we know, the team has only worked with gold nanoparticles, but of course, this investigation holds promising devlopements in a bunch of other fields like materials science, photonics, electronics and catalysis. Now . . . for the answers you've been waiting for: how exactly does this whole DNA and nanoparticles forming crystals work? Well, we know we like gems and beautiful minerals alike them - but why? How does their beauty come to exist? Listen up, because here are the juicy details: single crystals follow a certain crystal lattice that continues without interruption throughut and for this reason, there is an absence of defects, giving the crystals and thus in extension, minerals (gemstones) very unique properties optically, mechanically and electrically. Though one of the most significant conclusions from the experiment is that the ratio of DNA's linker's length to the nanoparticle's size is crucial during the transformation of a crystal from disorder to order. Additionally, it is when and only when the ratio is "right" will a perfect crystal form. I find the concept intriguing, that even the slightest difference could effect a crystal's entire structure; instead of a crisp, crystalline shape, all you're going to get is a sphere. The idea is to attach complementary DNA to the nanporaticles and then when they're being combined together, the particles will "find" each other and form a bond, assembling into a 3-D crystal. Nevertheless, in order to realize this, about 1 million nanoparticles in water were heated to a temperature slightly above the DNA linkers' melting point. Afterwards, the solution was cooled slowly back to regular room temperature over the course of two to three days. This is critical because the slow cooling allows single-stranded DNA to find its complement with the amount of given time and energy which ultimately, produces the high-quality crystal. The following animation should give you an idea how the single-stranded DNA has to find its complementary base pairs, which is a similar concept to what had happened during the experiment with the nanoparticles Simply breathtaking. Everything about it is amazing. The future, you ask? "There's no reason we can't grow extraordinarily large single crystals in the future using modifications of our technique," said Mirkin. Though admitedly, growing naturally-occuring minerals from crystals is without a doubt, tedious. But, if it brings success and continous industrial benefits in the future, this attempt to manipulate aspects of nature to produce even more of what is has already offered makes me wonder whether mining and digging up the Earth would even be necessary anymore since an alternative has been generated. This may be a significant discovery, but we should never forget that there's still so much, too much, in fact, in ONE SMALL WORLDexisting in ONE BIG UNIVERSEto uncover.
1 Comment
Jerry Liu
12/7/2016 05:48:02 am
Great post! The pictures and gifs are very helpful for visualizing crystalline structures on an atomic level. The tone of the post was also kept friendly and laidback but still informative. I like how you end every post with the same saying. Good job!
Reply
Leave a Reply. |
|